Davi Mendes Luna1; Ludmila Cristina Camilo Furtado1; Paulo Suassuna2; Mariana Meira Dolfini Silva3
DOI: 10.17545/eOftalmo/2025.0015
ABSTRACT
This study aimed to describe the management and prognosis of a young man with myotonic dystrophy type 1 and low vision in his right eye associated with subretinal neovascular membrane. The patient was 39 years old. Retinal imaging showed subretinal hemorrhage in the macular area, with exudation in the right eye. Optical coherence tomography showed a normoreflective and irregular vitreoretinal interface; the presence of hyporeflective material, indicating subfoveal fluid; and the presence of hyperreflective material revealing subfoveal fibrosis. Subretinal neovascular membrane was diagnosed in the patient. Clinical management involved intravitreal injections of ranibizumab. Optical coherence tomography revealed a normoreflective vitreoretinal interface, presence of foveal depression, absence of subfoveal fluid and intraretinal cysts, and presence of irregularities in the photoreceptor layer. The findings in this case confirmed the relationship between retinal changes in patients with myotonic dystrophy type 1 and their management.
Keywords: Myotonic dystrophy; Subretinal neovascular membrane; Ophthalmopathy; Visual acuity; Case report.
RESUMO
Objetivamos descrever o manejo e prognóstico de um jovem com distrofia miotônica tipo 1 e baixa visão associada à membrana neovascular sub-retiniana. Trata-se de um homem, 39 anos, portador de distrofia miotônica tipo 1 e baixa visão em olho direito. O mapeamento da retina mostrou hemorragia sub-retiniana em área macular, com exsudação em olho direito. Tomografia de coerência óptica mostrou interface vítreo-retiniana normorrefletiva e irregular, presença de material hiporrefletivo, compatível com líquido subfoveal e presença de material hiperrefletivo, revelando fibrose subfoveal. Foi diagnosticada membrana neovascular sub-retiniana. Isto foi seguido por injeções intravítreas de Ranibizumabe. A tomografia de coerência óptica revelou interface vitreorretiniana normorrefletiva, depressão foveal presente, ausência de líquido subfoveal e cistos intrarretinianos e presença de irregularidade na camada fotorreceptora. Com isso, a relação entre alterações retinianas em pacientes com distrofia miotônica tipo 1 é elucidada, além de auxiliar no manejo dessas alterações.
Palavras-chave: Distrofia miotônica; Membrana neovascular subretiniana; Oftalmopatia; Acuidade visual; Relatos de casos.
INTRODUCTION
Myotonic dystrophy type 1 (DM1), or Steinert disease, is a neuromuscular and multisystem genetic disorder1 caused by the abnormal CTG trinucleotide repeat in the DMPK gene2. The link between DM1 and various ophthalmic findings, such as ptosis, cataracts, and reduced intraocular pressure, is well known1. Retinal changes have also been reported in DM1, including alterations in the retinal pigment epithelium (RPE) and the existence of an epiretinal membrane3-5; however, data on the emergence of subretinal neovascular membrane (SRNVM) as a consequence of DM1 are scarce. This study aimed to describe the management and prognosis of a young man diagnosed with DM1 and low vision in the right eye (OD) associated with the development of SRNVM.
CASE REPORT
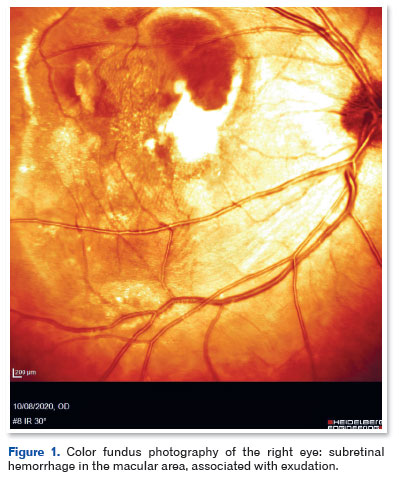
A 39-year-old man diagnosed with DM1 in early childhood (through a PCR test with capillary electrophoresis and Southern blot that detected pathological repeats of the CTG trinucleotide in DMPK) reported recent vision loss in the OD. On ophthalmologic examination, corrected visual acuity (CVA) was 20/100 in the OD and 20/20 in the OE. Biomicroscopy showed clear conjunctiva, transparent cornea, no reaction in the anterior chamber, and clear lens in both eyes. Extrinsic eye movement was preserved in both eyes. Retinal imaging showed subretinal hemorrhage in the macular area, associated with exudation in the OD (Figure 1). No retinal changes were observed in the OS. The findings of fluorescein angiography were hypofluorescence due to blockage in the macular area of the OD. Optical coherence tomography (OCT) revealed a normoreflective and irregular vitreoretinal interface, absence of foveal depression, presence of hyporeflective material indicating subfoveal fluid, and presence of hyperreflective material, suggesting subfoveal fibrosis (Figure 2).
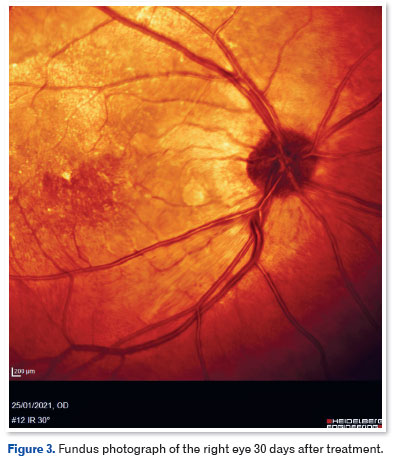
After being diagnosed with SRNVM in the OD, the patient received three intravitreal injections of ranibizumab 10 mg/mL (0.1 mL), 4 mm from the limbus, 30 days apart. He progressed with a significant improvement in visual acuity (VA): at the review 30 days after the last injection, VA was 20/40 in the OD. At this time, OCT showed a normoreflective vitreoretinal interface, presence of foveal depression, absence of subfoveal fluid, absence of intraretinal cysts, and presence of irregularity in the photoreceptor layer (Figures 3 and 4).
DISCUSSION
DM1 is an autosomal dominant disorder first described over a century ago, and several studies are increasingly focusing on the genetic mechanism underlying its progression2. Its cause, as described above, is the expansion of a tandem repeat1,2. There is a broad spectrum of clinical presentations that begin in the second, third, or fourth decade of life in 75% of cases. The most common first symptom of DM1 is myotonia2.
A series of ophthalmologic findings have been described in patients with DM11,2,4,6. Ikeda et al.6 reported that approximately 90% of their cohort had a history of cataracts or phacoemulsification. The second most frequent DM1 symptom is eyelid ptosis. Our patient did not present any of these symptoms, which makes the case more relevant.
In another study, the retina was evaluated in a cohort of patients with DM1 and compared with that of healthy individuals. They reported that patients with DM1 exhibited a greater central macular thickness, a higher prevalence of epiretinal membrane, and a greater number of butterfly-shaped and reticular pigmentary anomalies. The study correlated inner and outer retinal changes with increasing age and reported premature retinal aging in individuals with DM14.
Notably, macular degeneration due to advanced age is a main cause of retinal neovascularization, which was not a causal factor in the case described herein because the patient is a young adult7. Other associated causes of retinal neovascularization include uveitis, eye trauma, tumors, and hypertensive choroidopathy, which were absent in the patient's history. Thus, possible causes of the neovascular membrane were excluded, and DM1 was proposed as a probable causal factor8.
Subretinal neovascular processes occur in diseases affecting the outer retina and Bruch's membrane, and the current first-line treatment involves neutralizing vascular endothelial growth factor (VEGF), an important molecule in the pathogenesis of SRNVM9. The condition of the patient described herein improved satisfactorily with the use of ranibizumab, which inhibits the action of VEGF-A. The efficacy of ranibizumab has been assessed in other studies on patients with idiopathic choroidal neovascularization. Resorption of macular edema and significant improvement in VA were observed after an average of three injections per patient10.
The relevance of the present case to the current literature is noteworthy, given that the authors found limited descriptions of similar cases across major databases. Thus, this is a rare report describing a potential relationship between SRNVM and DM1, contributing substantially to the medical research.
In conclusion, cases such as the one described herein have not been found in the current literature. Reports of such cases might elucidate the relationship between retinal changes in patients with DM1 and the management of subsequent disorders, especially SRNVM.
ACKNOWLEDGMENTS
We thank the Oftalmax clinic for providing the structure and support for the study.
REFERENCES
1. Majid Moshirfar, Webster CR, Seitz TS, Ronquillo YC, Hoopes PC. Ocular Features and Clinical Approach to Cataract and Corneal Refractive Surgery in Patients with Myotonic Dystrophy. Clin Ophthalmol. 2022 Aug 25;16:2837-42.
2. Thornton CA. Myotonic Dystrophy. Neurol Clin. 2014;32(3):705-19.
3. Kirkegaard-biosca E, Berges-Marti M, Azarfane B, Cilveti E, Distefano L, García-Arumí J. Fundus flavimaculatus-like in myotonic dystrophy: a case report. BMC Ophthalmol. 2021; 21(1):240.
4. Abed E, Guglielmo D'Amico, Rossi S, Perna A, Laura M, Silvestri G. Spectral domain optical coherence tomography findings in myotonic dystrophy. Neuromuscul Disord. 2020;30(2):144-150.
5. Zinkernagel MS, Hornby SJ, MacLaren RE. Choroidal new vessels in type 1 myotonic dystrophy-related macular dystrophy respond to anti-VEGF therapy. Eye (Lond). 2012;26(12):1595-6.
6. Ikeda KS, Iwabe-Marchese C, França Jr MC, Nucci A, Carvalho KM de. Myotonic dystrophy type 1: frequency of ophthalmologic findings. Arq Neuropsiquiatr. 2016;74(3):183-8.
7. Weber ML, Heier JS. Choroidal Neovascularization Secondary to Myopia, Infection and Inflammation. Dev Ophthalmol. 2016;55:167-75.
8. Baxter SL, Pistilli M, Pujari SS, Liesegang TL, Suhler EB, Thorne JE, et al. Risk of choroidal neovascularization among the uveitides. Am J Ophthalmol. 2013;156(3):468-477.e2.
9. Campochiaro PA. Molecular pathogenesis of retinal and choroidal vascular diseases. Prog Retin Eye Res. 2015 Nov;49:67-81.
10. Chrapek O, Vostrovská Z, Šínová I, Chrapková B. Treatment of idiopathic choroidal neovascular membrane with ranibizumab - our experience. Cesk Slov Oftalmol. 2019;75(1):25-29.
| AUTHO'S INFORMATION |
|
 |
» Davi Mendes Luna https://orcid.org/0000-0002-3500-4621 http://lattes.cnpq.br/8536331220140020 |
 |
» Paulo Suassuna https://orcid.org/0006-5601-2934 http://lattes.cnpq.br/0322257309114269 |
 |
» Ludmila Cristina Camilo Furtado https://orcid.org/0000-0002-5497-4033 http://lattes.cnpq.br/9383771739969144 |
 |
» Mariana Meira Dolfini Silva https://orcid.org/0009-0008-9408-8168 http://lattes.cnpq.br/4918756386818033 |
Funding: The authors declare no funding.
Ethics committee approval: HUOC Hospital Complex/PROCAPE (CAAE: 83426624.6.0000.5192).
Conflicts of interest: The authors declare no conflicts of interest.
Received on:
January 10, 2025.
Accepted on:
September 24, 2025.